Proper nervous system function relies on small junctions between neurons that allow for communication. These are called synapses. In many organisms, the structural and molecular properties of synapses also underlie the mechanisms for memory encoding. However, the precise way synapses are assembled remains unclear. General models of synapse assembly (synaptogenesis) support the idea that connections between pre and post synaptic sites are an initiating event for synaptogenesis. Some data show, however, that protein organization at pre and post synaptic sites can occur independently of these trans-synaptic connections.
Neurexins are a family of cell-adhesion molecules (CAMs) that are known to promote trans-synaptic binding at presynaptic sites. As mentioned above, the prevailing idea regarding neurexins and other CAMs is that their trans-synaptic binding promotes synaptogenesis. Yet, there is a growing body of data from different animal models that shows neurexins are organized through intracellular interactions. Similarly, some synaptic assemblies and receptors appear to be pre-arranged prior to trans-synaptic connections. This raises the possibility that synaptic assembly may be internally organized, and CAMs may instead be involved in synaptic maturation. Frankel et al. set out to resolve some of these discrepancies using neurexin assembly as a guide.
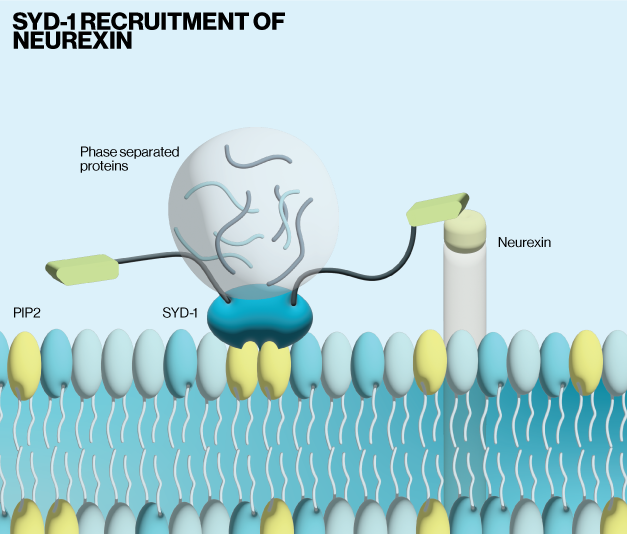
The authors first use a combination of in vivo imaging and mutagenesis to show that neurexin is capable of clustering at synaptic sites without extracellular connections. This led them to perform a screen of genes that altered neurexin localization in synapses. Their results identified the scaffolding protein SYD-1 as being positively correlated with neurexin clustering. Subsequent imaging of neurexin and SYD-1 together showed that they are tightly associated in synaptic zones. SYD-1 is predicted to have domains that target it to cell membranes and allow for lipid interactions. The authors showed via molecular dynamics simulations and in vitro binding assays (Echelon, P-6001) that SYD-1 binds PI(4,5)P2. Follow up experiments revealed that SYD-1 recruitment to the membrane in cells requires PI(4,5)P2, where it is then stabilized by liquid-liquid phase separated proteins to facilitate clustering of neurexin.
Synapse assembly and maturation has been implicated in numerous neurodevelopmental disorders, and neurexins in particular have been associated with schizophrenia and autism spectrum disorders. The current data provide new mechanistic detail to models of synapse development and may be informative for approaching new therapies for neuronal disorders.
Read the full article here:
Intracellular protein-lipid interactions drive presynaptic assembly prior to neurexin recruitment
Neuron (2025)
