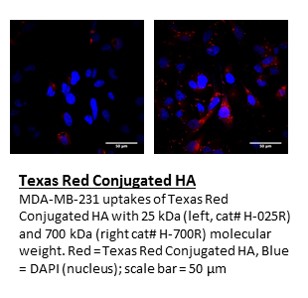
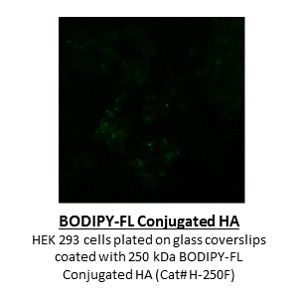
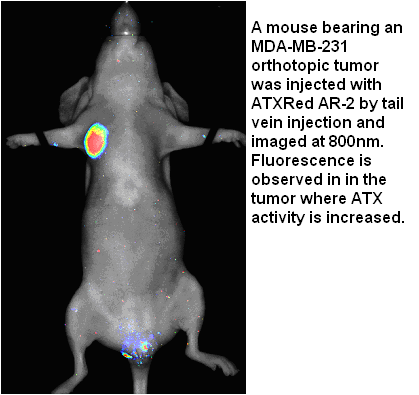
Sodium Hyaluronate is the sodium salt of hyaluronic acid (HA), also known as hyaluronan, a glycosaminoglycan consisting of D-glucuronic acid and N-acetyl-D-glucosamine disaccharide units. HA is one of several glycosaminoglycan components of the extracellular matrix of connective tissue. HA is a naturally occurring biopolymer involved in numerous biological processes including tissue hydration and structural scaffolding. HA is increasingly used as a reagent and investigated in medical, pharmaceutical and bioengineering applications. Its use as a reagent includes hydrogels for use in aesthetics, ophthalmology, rheumatology, urology, wound healing, and 3D bioprinting.
Our medical grade Sodium Hyaluronate is produced by HTL by fermentation of a Streptococcus equi strain (Group C of the Lancefield Classification / Non GMO / Without any material from animal origin). HTL’s proprietary process allows the production of Sodium Hyaluronate fiber with an exceptionally low level of impurities. HTL Sodium Hyaluronate raw material is a medical grade pharmaceutical product manufactured under cGMP conditions and is covered by a Certificate of Suitability of Monographs if the European Pharmacopoeia (CEP) and Drug Master File (DMF).
HTL Sodium Hyaluronate is aliquoted and distributed by Echelon for research use only.
MW range: 1300-1700 kDa (see Certificate of Analysis for lot specific MW)
Storage: dry product at 5 °C, protected from light and humidity. Solutions should be stored frozen at -20 °C or below.
About HTL: HTL is the world leader in the production of pharmaceutical grade Sodium Hyaluronate by fermentation. www.htlbiotech.com
Bulk discounts available, please email echelon@echelon-inc.com for information.
References
1) M.A. Serban, A. Skardal (2018) “Hyaluronan chemistries for three-dimensional matrix applications” Matrix Biology, 78-79, 337-3454. doi: 10.1016/j.matbio.2018.02.010.
2) C.B. Highley, G.D. Prestwich GD, Burdick JA. (2016) “Recent advances in hyaluronic acid hydrogels for biomedical applications.” Curr Opin Biotechnol. 40, 35-40. doi: 10.1016/j.copbio.2016.02.008.
3) J.A. Burdick, G.D. Prestwich (2011) “Hyaluronic acid hydrogels for biomedical applications.” Adv Mater. 23, H41-56. doi: 10.1002/adma.201003963.
4) A. Dodero, R. Williams, et al. (2019) “A micro-rheological and rheological study of biopolymers solutions: Hyaluronic acid” Carbohydrate Polymers, 203, 349-355, doi:10.1016/j.carbpol.2018.09.072.
5) P.A. Simmons, J.G. Vehige (2017) “Investigating the potential benefits of a new artificial tear formulation combining two polymers.” Clin Ophthalmol. 11, 1637-1642. doi:10.2147/OPTH.S135550.